上皮性卵巢肿瘤(epithelial ovarian tumor,EOT)占原发性卵巢肿瘤50%~70%,起源与发病机制长期以来存在争议,按生物学行为分为良性、交界性和恶性[1]。恶性EOT,即上皮性卵巢癌(epithelial ovarian cancer,EOC),约占卵巢癌的90%, 起病隐匿,缺乏有效的早期诊断方法,近年来发病率呈上升趋势,多在III期(51%)和IV期(29%)被诊断[2],容易错过最佳治疗时机导致死亡率增加,5年生存率低于30%[3-4],如果能早期发现并采用标准手术和辅助疗法进行治疗,则5年生存率高达90%,死亡率可以降低10%~30%[5-6]。因此术前用高度敏感的生物标志物来辅助诊断EOC,使患者及时接受标准化治疗可显著改善预后。交界性上皮性卵巢肿瘤(borderline epithelial ovarian tumor,BOT)生物学行为处于良性和恶性之间,与EOC主要区别是缺乏间质浸润和快速浸润发展,预后较EOC好,5年生存率达95%~100%[7-8]。目前很多研究支持BOT可能是相应组织学类型EOC的前体病变的假设。然而,BOT和EOC临床症状相似,二者缺乏特异性标志物和影像学检查手段,术前诊断容易混淆,术中冰冻存在误差,这为后续手术方案的制定带来了困难。因此寻找术前易于获得并能辅助鉴别BOT和EOC的潜在检测方法至关重要。
近年来,探索肿瘤发生发展的相关炎症理论被广泛研究。术前炎症细胞计数及其衍生比值[9-14]已被认为是各种恶性肿瘤最具诊断潜力的替代生物指标。纤维蛋白原和白蛋白比值(fibrinogen to albumin ration, FAR)最近被确定为影响各种恶性肿瘤预后的因素,血清纤维蛋白原升高和白蛋白水平降低被认为是检测全身炎症反应的有效生物标志物[15]。癌症抗原125(carbohydrate antigen 125, CA125)和人附睾蛋白4(human epididymal protein 4, HE4)是诊断EOT最常用的血清生物标志物,但敏感性和特异性有限。卵巢恶性肿瘤风险预测模型(risk of ovarian malignacy algorithm, ROMA)指数将CA125、HE4水平与绝经状态相结合来评估盆腔包块的恶性风险,提高了CA125、HE4检测EOC特异性[16]。超声检查是卵巢肿瘤筛查和术前诊断的首选影像学检查,操作方便、普及率高,可提高检测特异性。很多研究探讨了肿瘤标志物和(或)炎症指标在区分良恶性卵巢肿瘤中的诊断价值,而在交界性卵巢肿瘤术前诊断中的价值很少被提及。本研究目的是回顾性分析BOT和EOC术前血清中炎症指标、凝血指标、肿瘤标志物及超声特征的差异,探讨术前血清指标联合超声特征对BOT和EOC的鉴别诊断价值,寻找联合检测方法增加二者术前诊断准确率以期为临床诊断提供参考。
对象与方法
一、研究对象
选取2018年8月—2023年12月于山西医科大学第一医院接受手术且术后病理证实为BOT和EOC的患者各112例作为研究对象,收集其临床资料进行回顾性分析。
纳入标准:(1)入院后诊断为BOT和EOC,且初次行手术治疗的患者;(2)术后病理证实,可按FIGO分期;(3)完整的病例资料:术前1周内血清化验、超声检查和病理资料及一般临床资料完善。排除标准:(1)合并严重心肝肾疾病、自身免疫性疾病、血液系统疾病、结缔组织病、血栓性疾病、结核病;(2)非上皮性卵巢肿瘤、其他部位恶性肿瘤或肿瘤转移;(3)接受过放疗和化疗等治疗;(4)术前两周有急性感染、使用抗生素、输血、接受抗凝治疗(口服抗凝剂、乙酰水杨酸、他汀类药物等);(5)术前3个月接受过腹部除卵巢外的其他手术;(6)经期、妊娠、吸烟、饮酒;(7)病例资料不完整。
二、研究方法
1.数据收集:(1)术前血清化验。所有患者均于术前一周内空腹状态下抽取肘静脉血,检测血常规、凝血系列、白蛋白、肿瘤标志物。炎症指标包括血小板(platelet, Plt)、淋巴细胞(lymphocyte, L)、中性粒细胞计数(neutrophil, N)、单核细胞计数(monocyte, M)、血小板/淋巴细胞比值(platelet to lymphocyte ratio, PLR)、中性粒细胞/淋巴细胞比值(neutrophil to lymphocyte ratio, NLR)、单核细胞/淋巴细胞比值(monocyte to lymphocyte ratio, MLR)、血小板/中性粒细胞比值(platelet to neutrophil ratio, PNR)、全身免疫炎症指数(systemic immune inflammatory index, SII)、全身炎症反应指数(systemic inflammatory response index, SIRI)。凝血指标包括D-二聚体(D-dimer, D-d)、纤维蛋白原(fibrinogen, Fib)、白蛋白(albumin, ALB)及FAR。肿瘤标志物包括CA125、HE4以及ROMA。(2)术前超声检查。所有病例均行经阴道或经肛门彩色多普勒超声检查,记录超声特征包括有无乳头状突起、血流信号、间隔、质地及肿瘤最大径线等。(3)病理资料。由两名资深病理学家对病理进行审查。
2.指标计算:绝经前预测指数(predictive index)=-12.0+2.38×LN(HE4)+0.0626×LN(CA125);绝经后 PI=- 8.09 + 1.04 ×LN(HE4)+0.732 ×LN(CA125)。ROMA指数(%)=exp(PI)/[1+exp(PI)]×100,exp(PI)=ePI,exp为指数函数。
3.统计分析方法:使用IBM SPSS软件V25.0版本对数据进行分析。定量资料:符合正态性、方差齐性数据采用t检验,表示为![]() 非正态性数据采用秩和检验,表示为M(P25, P75)。定性资料表示为例(%),两组间差异性分析使用χ2检验或Fisher确切概率法。分析鉴别BOT和EOC的指标采用二元Logistic回归分析,单因素分析P<0.1认为差异有统计学意义[17],多因素分析P<0.05认为差异有统计学意义。采用ROC曲线比较鉴别对BOT和EOC的诊断性能。
非正态性数据采用秩和检验,表示为M(P25, P75)。定性资料表示为例(%),两组间差异性分析使用χ2检验或Fisher确切概率法。分析鉴别BOT和EOC的指标采用二元Logistic回归分析,单因素分析P<0.1认为差异有统计学意义[17],多因素分析P<0.05认为差异有统计学意义。采用ROC曲线比较鉴别对BOT和EOC的诊断性能。
结果
一、BOT和EOC术前血清学指标及超声特征的差异性分析
1.两组患者术前血清指标差异性分析:两组患者的Plt、L、NLR、PLR、MLR、SII、SIRI、D-d、Fib、ALB、FAR、HE4、CA125、ROMA血清水平差异有统计学意义(P<0.05);N、M、PNR等指标的血清水平差异无统计学意义(P>0.05),见表1。
表1 BOT组和EOC组的术前血清指标比较
Table 1 Comparison of preoperative serum indexes between BOT group and EOC group
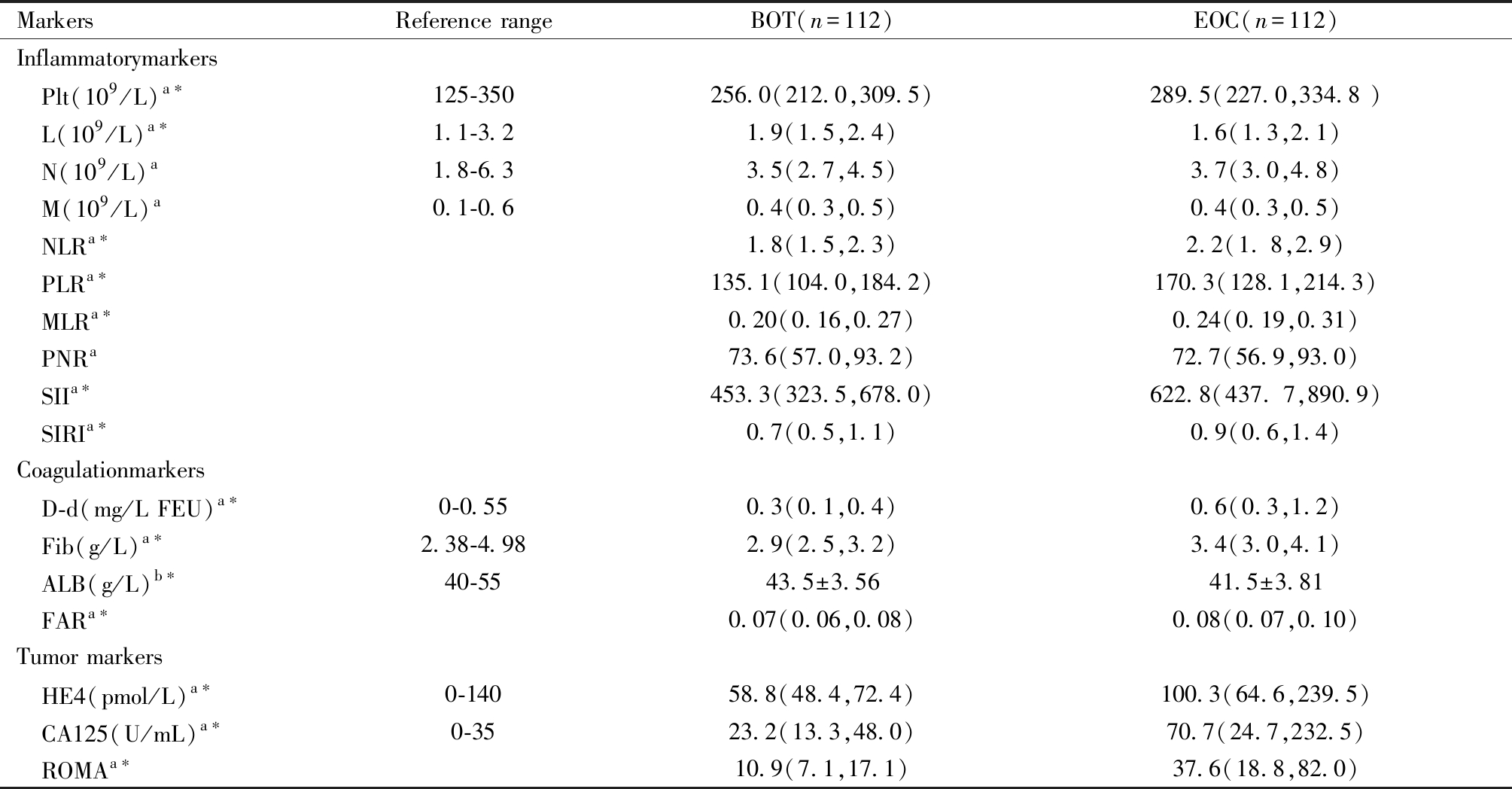
Note: BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; a:Non-normality of quantitative data, M(P25,P75); b:Normality of quantitative ![]() *Comparison among groups,P<0.05.
*Comparison among groups,P<0.05.
MarkersReference rangeBOT(n=112)EOC(n=112)Inflammatorymarkers Plt(109/L)a∗125-350256.0(212.0,309.5)289.5(227.0,334.8 ) L(109/L)a∗1.1-3.21.9(1.5,2.4)1.6(1.3,2.1) N(109/L)a1.8-6.33.5(2.7,4.5)3.7(3.0,4.8) M(109/L)a0.1-0.60.4(0.3,0.5)0.4(0.3,0.5) NLRa∗1.8(1.5,2.3)2.2(1. 8,2.9) PLRa∗135.1(104.0,184.2)170.3(128.1,214.3) MLRa∗0.20(0.16,0.27)0.24(0.19,0.31) PNRa73.6(57.0,93.2)72.7(56.9,93.0) SIIa∗453.3(323.5,678.0)622.8(437. 7,890.9) SIRIa∗0.7(0.5,1.1)0.9(0.6,1.4)Coagulationmarkers D-d(mg/L FEU)a∗0-0.550.3(0.1,0.4)0.6(0.3,1.2) Fib(g/L)a∗2.38-4.982.9(2.5,3.2)3.4(3.0,4.1) ALB(g/L)b∗40-5543.5±3.5641.5±3.81 FARa∗0.07(0.06,0.08)0.08(0.07,0.10)Tumor markers HE4(pmol/L)a∗0-14058.8(48.4,72.4)100.3(64.6,239.5) CA125(U/mL)a∗0-3523.2(13.3,48.0)70.7(24.7,232.5) ROMAa∗10.9(7.1,17.1)37.6(18.8,82.0)
2.两组患者超声特征差异性分析:超声特征中有无乳头、间隔、血流信号及质地在BOT组和EOC组中差异具有统计学意义(P<0.05),两组肿块最大径线及最大径线是否大于10cm无统计学差异(P>0.05)。见表2。
表2 BOT组和EOC组的超声特征比较
Table 2 Comparison of ultrasonographic characteristics between BOT group and EOC group
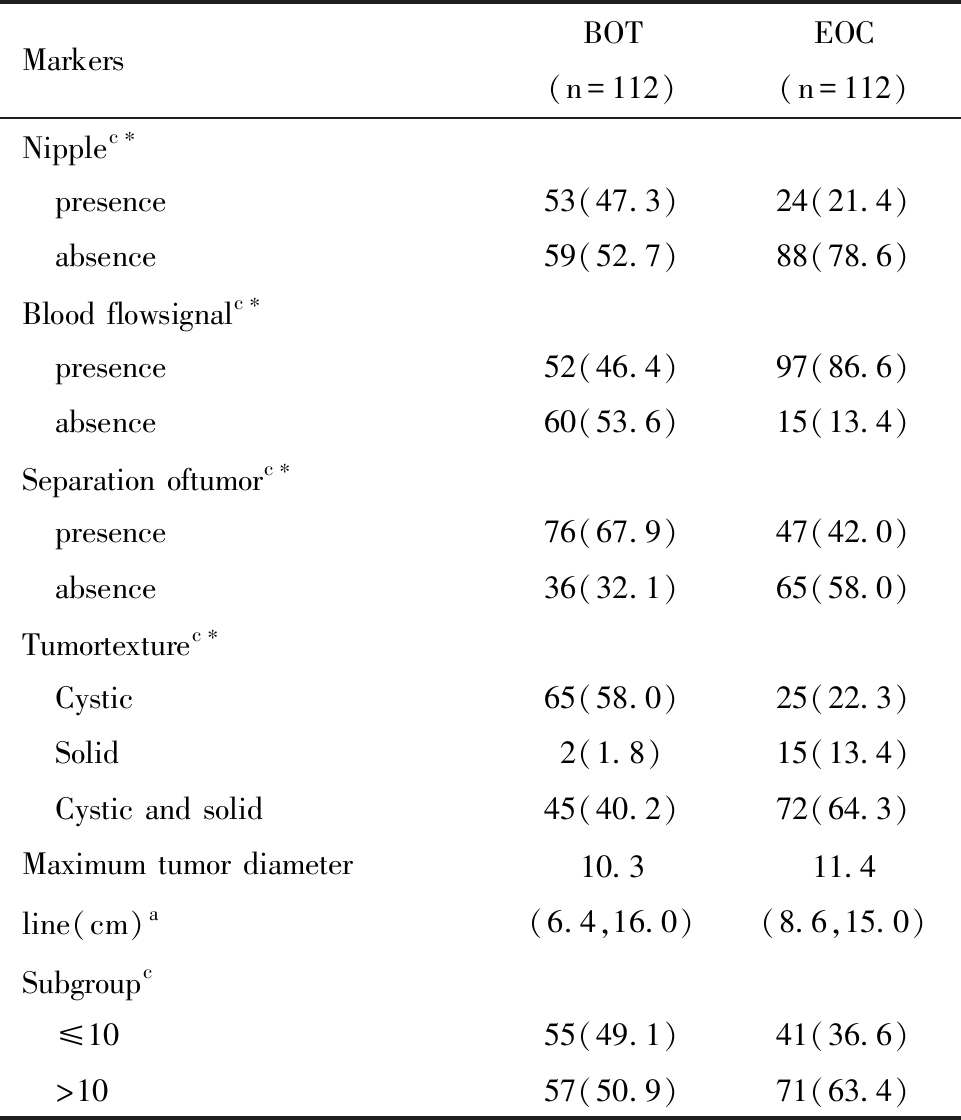
BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; a:Non-normality of quantitative data,M(P25,P75);c:Qualitative data, n(%); *Comparison among groups,P<0.05
MarkersBOT(n=112)EOC(n=112)Nipplec∗ presence53(47.3)24(21.4) absence59(52.7)88(78.6)Blood flowsignalc∗ presence52(46.4)97(86.6) absence60(53.6)15(13.4)Separation oftumorc∗ presence76(67.9)47(42.0) absence36(32.1)65(58.0)Tumortexturec∗ Cystic65(58.0)25(22.3) Solid2(1.8)15(13.4) Cystic and solid45(40.2)72(64.3)Maximum tumor diameterline(cm)a10.3(6.4,16.0)11.4(8.6,15.0)Subgroupc ≤1055(49.1)41(36.6) >1057(50.9)71(63.4)
3. HE4、CA125血清水平对BOT和EOC的鉴别诊断效能:以病理学诊断结果为金标准,阳性结果为病理确诊的112例EOC患者,阴性结果为病理确诊的112例BOT患者。血清HE4水平用于区分BOT和EOC的敏感度和特异度分别是78.8%(67/85)和67.6%(94/139)。血清CA125水平用于区分BOT和EOC的敏感度和特异度分别是64.9%(72/111)和64.6%(73/113)。见表3。
表3 HE4、CA125血清水平对BOT和EOC的鉴别诊断效能, n(%)
Table 3 Diagnostic efficacy of serum levels of HE4 and CA125 in differentiating BOT and EOC, n(%)

BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; c:Qualitative data, n(%); *Comparison among groups,P<0.05
MarkersBOT(n=112)EOC(n=112)HE4(pmol/L)c∗ ≤14094(83.9)45(40.2) >14018(16.1)67(59.8)CA125(U/mL)c∗ ≤3573(65.2)40(35.7) >3539(34.8)72(64.3)
二、BOT和EOC的二元Logistic回归分析
1.单因素分析:结果显示,超声特征中有无血流信号、乳头、间隔及质地、最大径线是否大于10 cm等差异有统计学意义(P<0.1);术前血清指标中Plt、L、PLR、NLR、MLR、SII、SIRI、D-d、Fib、ALB、FAR、CA125、HE4及ROMA等差异有统计学意义(P<0.1)。见表4。
表4 BOT组和EOC组的单因素分析
Table 4 Univariate analysis between BOT group and EOC group
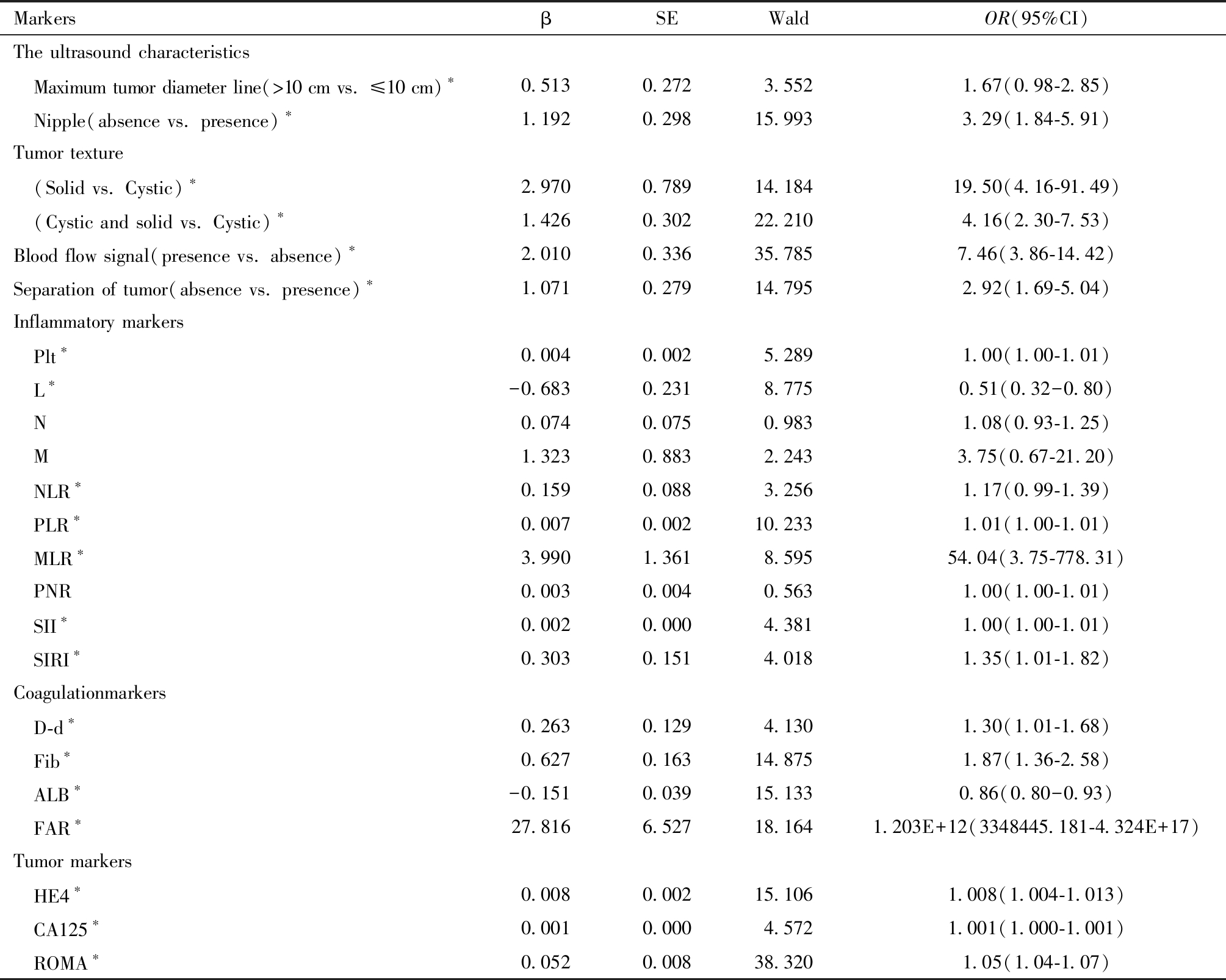
BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; *Comparison among groups,P<0.1. OR :odds ratio; 95% CI:95% confidential interval.
MarkersβSEWaldOR(95%CI)The ultrasound characteristics Maximum tumor diameter line(>10 cm vs. ≤10 cm)∗0.5130.2723.5521.67(0.98-2.85) Nipple(absence vs. presence)∗1.1920.29815.9933.29(1.84-5.91)Tumor texture (Solid vs. Cystic)∗2.9700.78914.18419.50(4.16-91.49) (Cystic and solid vs. Cystic)∗1.4260.30222.2104.16(2.30-7.53)Blood flow signal(presence vs. absence)∗2.0100.33635.7857.46(3.86-14.42)Separation of tumor(absence vs. presence)∗1.0710.27914.7952.92(1.69-5.04)Inflammatory markers Plt∗0.0040.0025.2891.00(1.00-1.01) L∗-0.6830.2318.7750.51(0.32-0.80) N0.0740.0750.9831.08(0.93-1.25) M1.3230.8832.2433.75(0.67-21.20) NLR∗0.1590.0883.2561.17(0.99-1.39) PLR∗0.0070.00210.2331.01(1.00-1.01) MLR∗3.9901.3618.59554.04(3.75-778.31) PNR0.0030.0040.5631.00(1.00-1.01) SII∗0.0020.0004.3811.00(1.00-1.01) SIRI∗0.3030.1514.0181.35(1.01-1.82)Coagulationmarkers D-d∗0.2630.1294.1301.30(1.01-1.68) Fib∗0.6270.16314.8751.87(1.36-2.58) ALB∗-0.1510.03915.1330.86(0.80-0.93) FAR∗27.8166.52718.1641.203E+12(3348445.181-4.324E+17)Tumor markers HE4∗0.0080.00215.1061.008(1.004-1.013) CA125∗0.0010.0004.5721.001(1.000-1.001) ROMA∗0.0520.00838.3201.05(1.04-1.07)
2.多因素分析:单因素分析中BOT组和EOC组最大径线是否大于10 cm的P值<0.1,符合多因素分析纳入标准,但与BOT组相比EOC组最大径线较大及最大径线大于10 cm的病例数较多(差异无统计学意义),不符合共识,因此多因素分析中不纳入最大径线是否大于10 cm这一指标。将有意义指标纳入多因素Logistic回归分析中,进行逐步选择变量分析,结果表明超声特征中有无乳头、间隔及血流信号是术前鉴别BOT和EOC的独立因素(P<0.05),术前血清指标中Plt、L、MLR、SII、Fib、ROMA是术前鉴别BOT和EOC的独立因素(P<0.05)。见表5。
表5 BOT组和EOC组的多因素分析
Table 5 Multivariate analysis between BOT group and EOC group
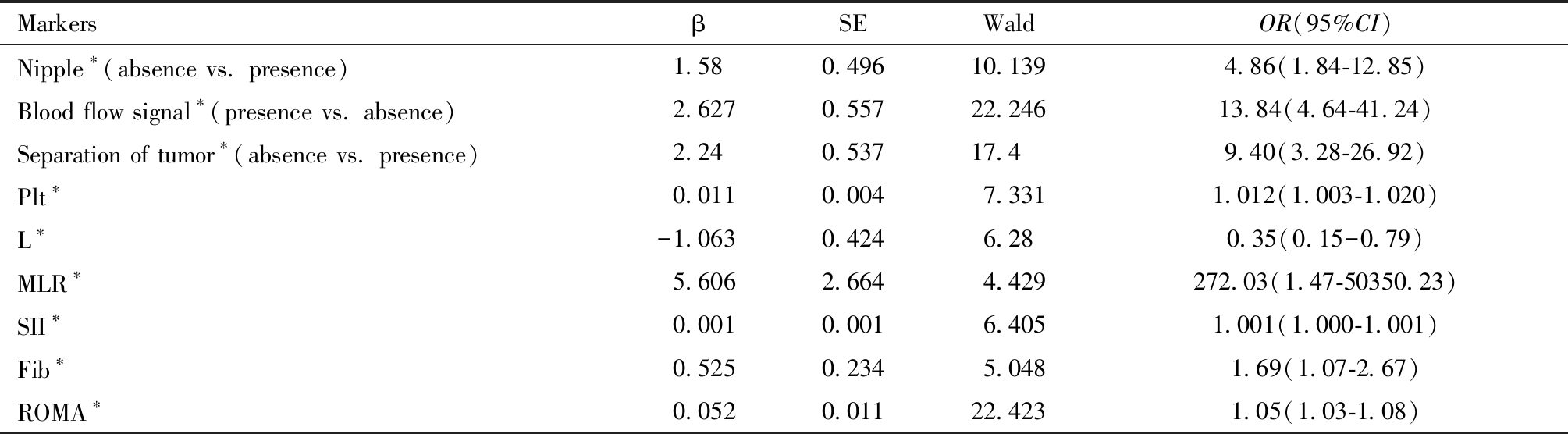
Note: BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; *Comparison among groups, P<0.05; OR :odds ratio; 95% CI:95% confidential interval.
MarkersβSEWaldOR(95%CI)Nipple∗(absence vs. presence)1.580.49610.1394.86(1.84-12.85)Blood flow signal∗(presence vs. absence)2.6270.55722.24613.84(4.64-41.24)Separation of tumor∗(absence vs. presence)2.240.53717.49.40(3.28-26.92)Plt∗0.0110.0047.3311.012(1.003-1.020)L∗-1.0630.4246.280.35(0.15-0.79)MLR∗5.6062.6644.429272.03(1.47-50350.23)SII∗0.0010.0016.4051.001(1.000-1.001)Fib∗0.5250.2345.0481.69(1.07-2.67)ROMA∗0.0520.01122.4231.05(1.03-1.08)
三、各项指标联合检测对BOT和EOC的鉴别价值
1.血清指标联合检测对BOT和EOC的鉴别价值:各项指标单独检测对鉴别诊断BOT和EOC均有一定的诊断效能(P<0.05),但鉴别价值不高(见表6和图1)。血清指标联合检测对BOT和EOC的鉴别诊断中,ROMA+SII+Fib+MLR+Plt+L联合检测的AUC最大且特异度和PPV最高,其余五项组合的敏感度和NPV均呈递增趋势(见表7和图2)。
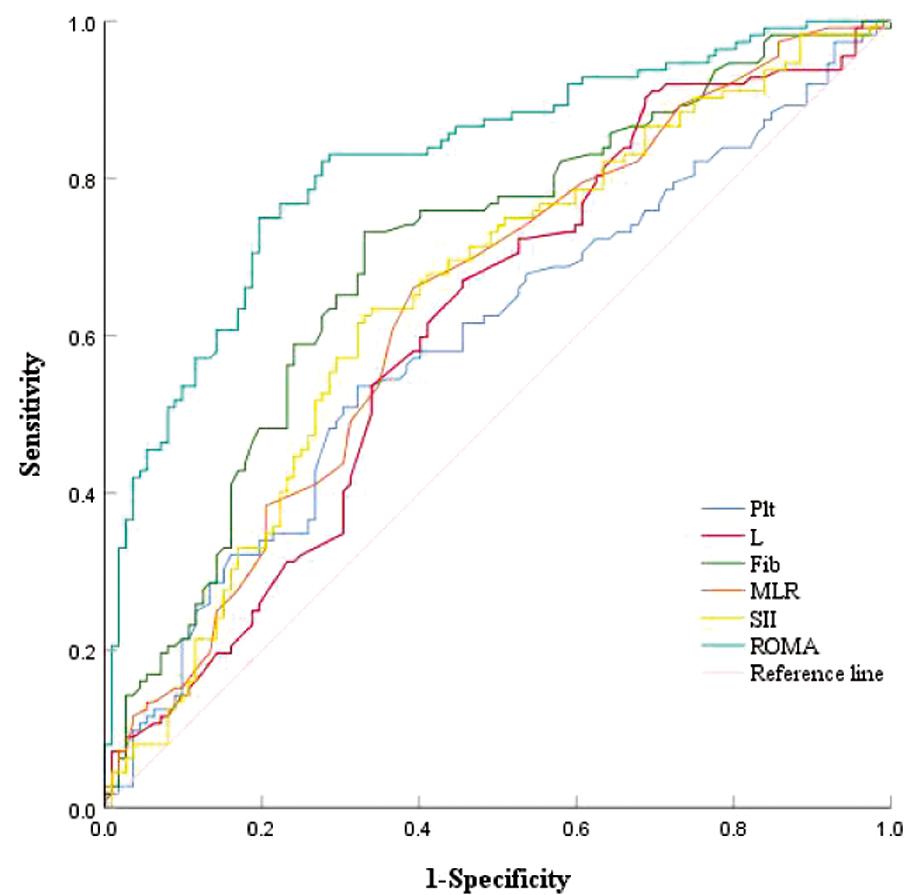
图1 各血清指标单独预测区分BOT和EOC的ROC曲线图
Figure 1 ROC curve of each serum index alone to predict BOT and EOC
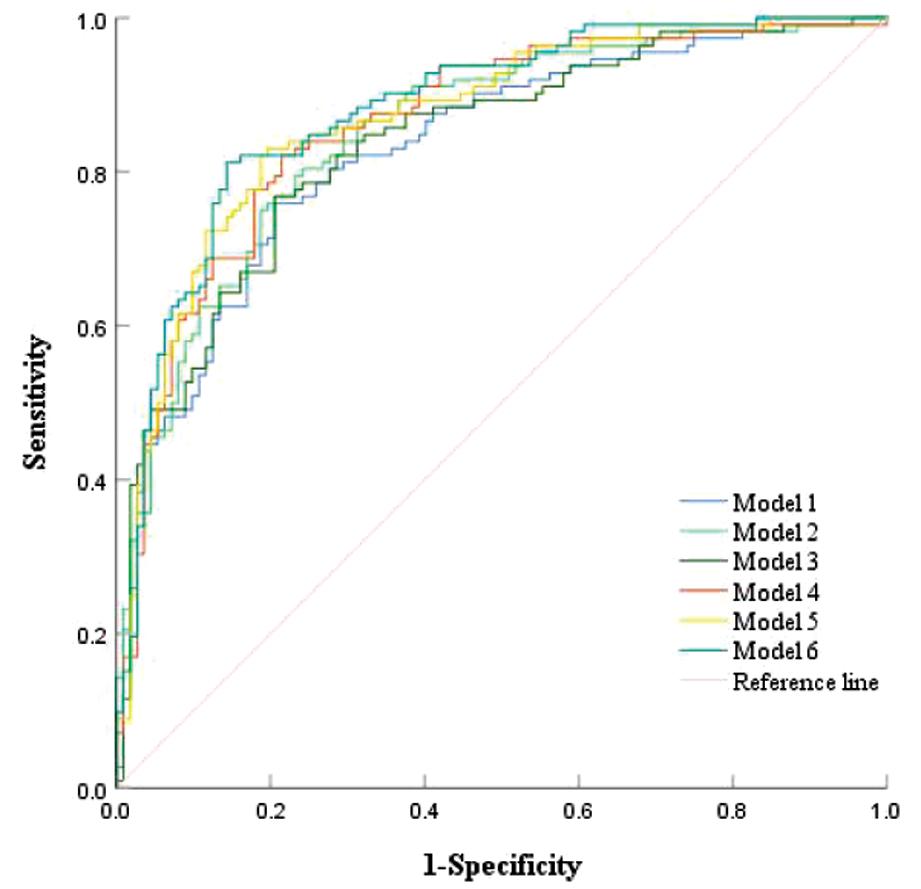
图2 血清指标联合预测区分BOT和EOC的ROC曲线图
Figure 2 ROC curve of different serum indexes combined to predict BOT and EOC
表6 各指标单独检测对BOT和EOC的鉴别价值
Table 6 Differential value of index detection alone in the diagnosis of BOT and EOC
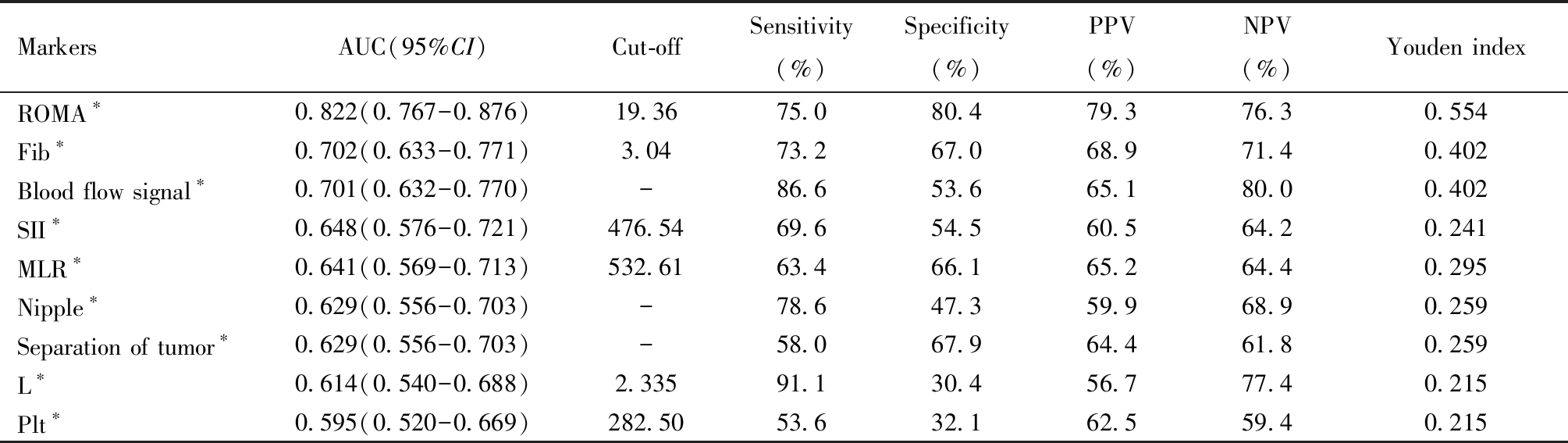
BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; *Comparison among groups,P<0.05; OR :odds ratio; 95% CI:95% confidential interval; PPV:positive predictive value; NPV:negative predictive value
MarkersAUC(95%CI)Cut-offSensitivity(%)Specificity(%)PPV(%)NPV(%)Youden indexROMA∗0.822(0.767-0.876)19.3675.080.479.376.30.554Fib∗0.702(0.633-0.771)3.0473.267.068.971.40.402Blood flow signal∗0.701(0.632-0.770)-86.653.665.180.00.402SII∗0.648(0.576-0.721)476.5469.654.560.564.20.241MLR∗0.641(0.569-0.713)532.6163.466.165.264.40.295Nipple∗0.629(0.556-0.703)-78.647.359.968.90.259Separation of tumor∗0.629(0.556-0.703)-58.067.964.461.80.259L∗0.614(0.540-0.688)2.33591.130.456.777.40.215Plt∗0.595(0.520-0.669)282.5053.632.162.559.40.215
表7 血清指标联合检测对BOT和EOC的鉴别价值
Table 7 Differential value of combined detection of serum indicators in the diagnosis of BOT and EOC
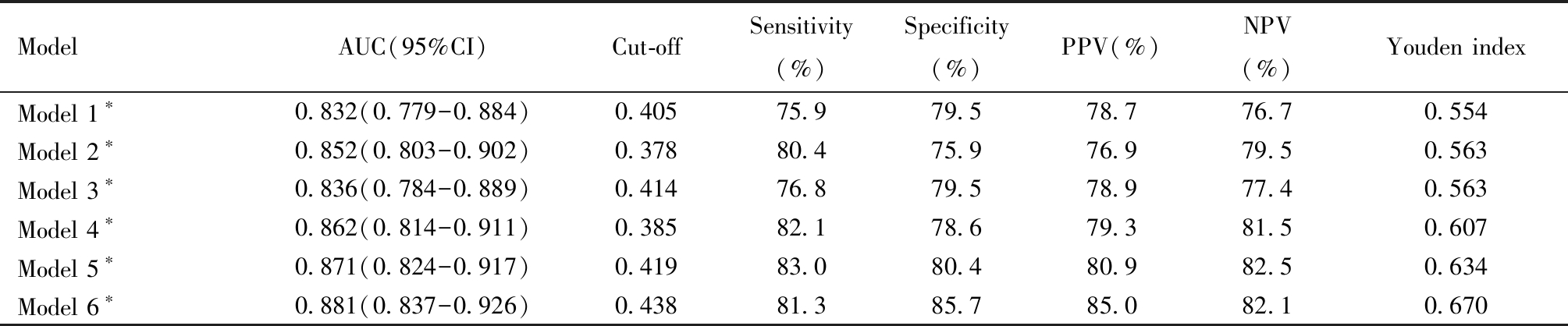
BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; *Comparison among groups, P<0.05. OR :odds ratio; 95% CI:95% confidential interval; PPV:positive predictive value; NPV:negative predictive value; Model 1: ROMA+SII; Model2: ROMA+SII+Fib ; Model 3:ROMA+SII+MLR; Model 4: ROMA+SII+Fib+MLR ; Model 5: ROMA+SII+Fib+MLR+Plt ; Model 6: ROMA+SII+Fib+MLR+Plt+L
ModelAUC(95%CI)Cut-offSensitivity(%)Specificity(%)PPV(%)NPV(%)Youden indexModel 1∗0.832(0.779-0.884)0.40575.979.578.776.70.554Model 2∗0.852(0.803-0.902)0.37880.475.976.979.50.563Model 3∗0.836(0.784-0.889)0.41476.879.578.977.40.563Model 4∗0.862(0.814-0.911)0.38582.178.679.381.50.607Model 5∗0.871(0.824-0.917)0.41983.080.480.982.50.634Model 6∗0.881(0.837-0.926)0.43881.385.785.082.10.670
2.各因素联合检测对BOT和EOC的鉴别价值:血清指标与超声特征联合检测对BOT和EOC的鉴别诊断中,联合4的AUC最大且敏感度和NPV最高,联合5的特异度和PPV最高,联合6很好地平衡了敏感度、特异度、PPV和NPV,且均高于85.0%,见表8和图3。
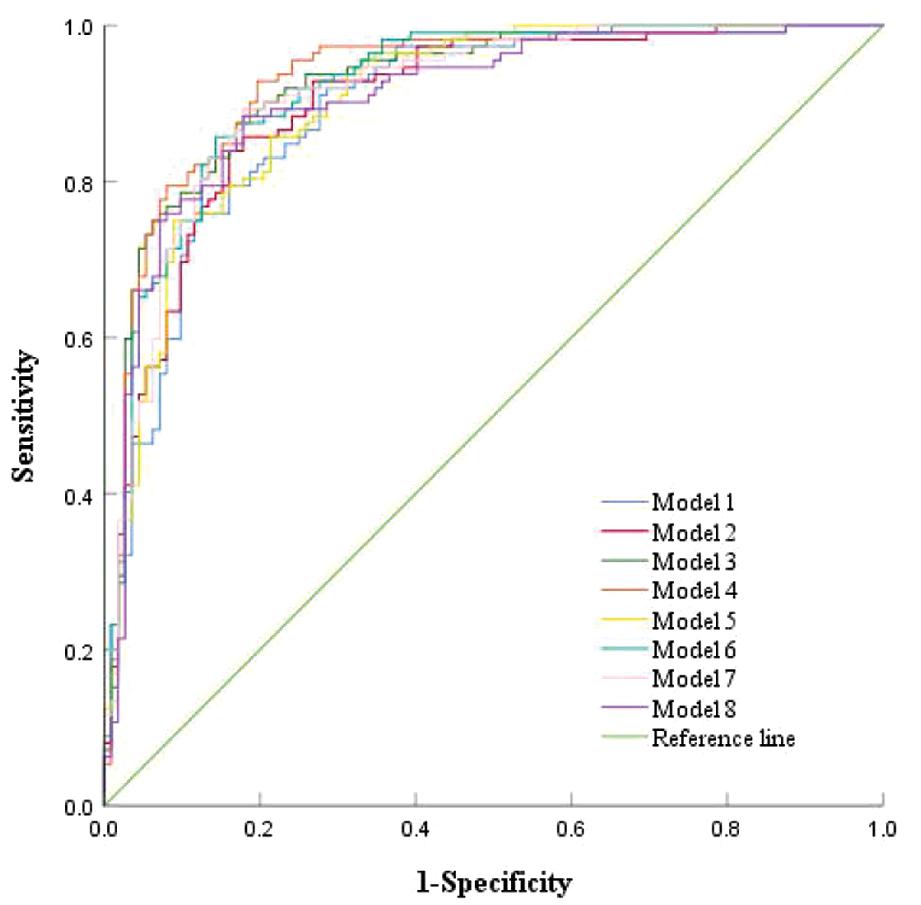
图3 各因素联合预测区别BOT和EOC的ROC曲线图
Figure 3 ROC curve of different serum and ultrasound indexes combined to predict BOT and EOC
Note: BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor
表8 各指标联合检测对BOT和EOC的鉴别价值
Table 8 Differential value of combined detection of indicators in the diagnosis of BOT and EOC
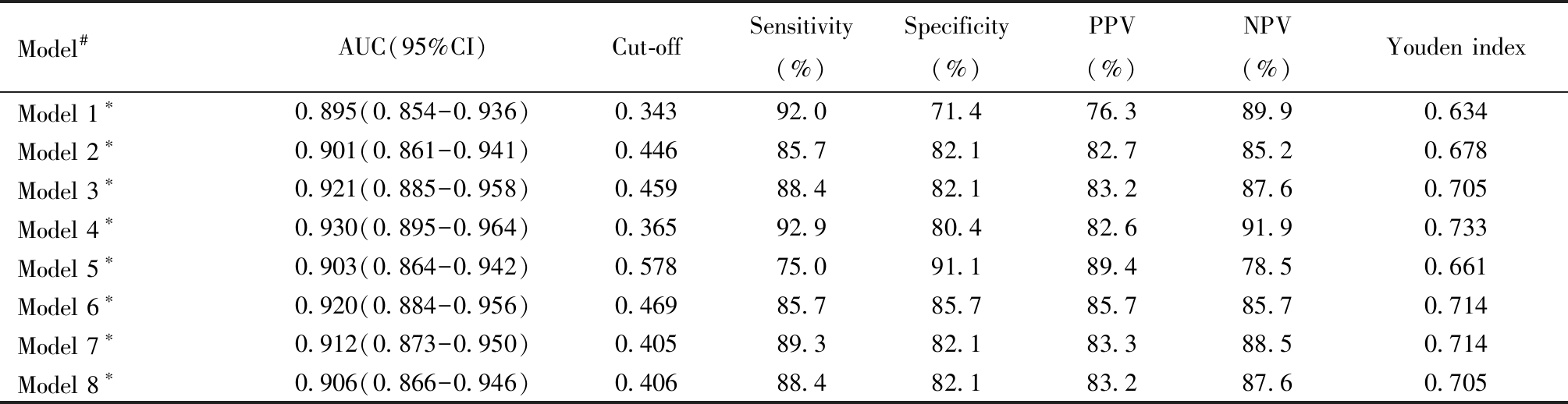
BOT: borderline ovarian tumor; EOC: epithelial ovarian tumor; *Comparison among groups,P<0.05. OR :odds ratio; 95% CI:95% confidential interval; PPV:positive predictive value; NPV:negative predictive value; # Model including: Model 1:ROMA+SII+Fib+MLR+Plt+ Blood flow signal; Model 2:ROMA+SII+Fib+MLR+Plt+ Blood flow signal + Nipple; Model 3:ROMA+SII+Fib+MLR+Plt+ Blood flow signal + Nipple + Separation of tumor; Model 4:ROMA+SII+Fib+MLR+Plt+L+ Blood flow signal + Nipple + Separation of tumor; Model 5:ROMA+SII+Fib+MLR+Plt+L+ Blood flow signal; Model 6:ROMA+SII+Fib+MLR+Plt+L+ Blood flow signal + Separation of tumor; Model 7:ROMA+SII+Fib+MLR+Plt+L+ Blood flow signal + Nipple; Model 8:ROMA+SII+Fib+MLR+Plt+L+ Nipple + Separation of tumor
Model#AUC(95%CI)Cut-offSensitivity(%)Specificity(%)PPV(%)NPV(%)Youden indexModel 1∗0.895(0.854-0.936)0.34392.071.476.389.90.634Model 2∗0.901(0.861-0.941)0.44685.782.182.785.20.678Model 3∗0.921(0.885-0.958)0.45988.482.183.287.60.705Model 4∗0.930(0.895-0.964)0.36592.980.482.691.90.733Model 5∗0.903(0.864-0.942)0.57875.091.189.478.50.661Model 6∗0.920(0.884-0.956)0.46985.785.785.785.70.714Model 7∗0.912(0.873-0.950)0.40589.382.183.388.50.714Model 8∗0.906(0.866-0.946)0.40688.482.183.287.60.705
讨论
BOT和EOC的临床症状相似,预后及生物学行为却不相同,故治疗策略存在差异。两者术前诊断主要依靠血清学检查及影像学检查,但均缺乏特异性。术中冰冻可为手术提供重要参考,却也有一定误差,如组织学类型、肿瘤大小、肿瘤偏侧性和病理专家水平等因素均会影响冰冻切片诊断的准确性[18-19],术中冰冻和术后石蜡病理在BOT中的总符合率为75%左右[20-22],术中冰冻对BOT与EOC鉴别准确率仅约51%[23]。EOC误诊为BOT时存在手术范围不足和延误治疗风险,BOT误诊为EOC时将导致不必要地扩大手术范围、增加患者手术损伤、甚至影响有生育要求患者的妊娠结局,因此术前鉴别诊断两者尤为重要。目前一些血清指标已被广泛研究,用于肿瘤的术前诊断及术后随访,联合检测研究也取得了显著成果,但将术前血清指标与超声特征联合应用于BOT和EOC鉴别诊断的研究较少,我们的研究表明术前血清指标Plt、L、MLR、SII、Fib、ROMA和超声特征中有无乳头、间隔、血流信号联合检测用于BOT和EOC的术前鉴别具有良好的诊断效能。
多项研究表明,炎症引起机体微环境的改变,肿瘤的发生、发展、侵袭、转移都伴随着炎症反应的变化[24-25]。目前有许多针对术前炎症细胞衍生比值如PLR、NLR、MLR、PNR、SII、SIRI的研究显示它们与多种恶性肿瘤的诊断和预后相关,如淋巴细胞单核细胞比值(LMR)对卵巢良性肿瘤和恶性肿瘤具有诊断价值[26];PLR可作为多种癌症的预测因子[27];NLR与上皮性卵巢癌的分期有关[28]。SII可代表体内不同的炎症和免疫途径[29],已被用于一系列恶性肿瘤的诊断和治疗[30-32],比NLR或PLR具有更强的预测能力。SIRI可作为NLR和LMR的综合指标,比三者单独时更能稳定地反映病情[33]。Huang等人[33]发现SIRI、PLR是术前预测良性和恶性卵巢肿瘤的有效指标,且EOC晚期组SIRI和PLR水平高于早期组。目前很少有研究关注SII、SIRI在术前鉴别EOC和BOT的作用,本研究显示EOC组中PLR、NLR、MLR、SII、SIRI水平均大于BOT组且差异具有统计学意义,多因素分析显示MLR、SII是术前鉴别BOT和EOC的独立因素,但两者的敏感度和特异度均较低(<70%),今后需收集更多的样本进一步研究。
既往研究发现CA125能够区分卵巢肿瘤组织和正常卵巢组织[34],但在卵巢癌早期敏感性较低,只有23%~50%有表达,特异性为78%,AUC也相对较低(0.78)[35],本研究中CA125用于区分BOT和EOC的敏感度和特异度分别是64.86%和64.60%。此外,如经期、妊娠、急性盆腔炎、子宫腺肌症、子宫肌瘤、子宫内膜异位症等均可导致CA125升高[36],因此它可能不是鉴别诊断BOT和EOC的理想标志物。HE4在卵巢癌组织中过度表达,特别是子宫内膜样卵巢癌,有研究发现HE4的特异性为86%,AUC(0.893)高于CA125(0.865) [37-38],本研究中HE4水平用于区分BOT和EOC的敏感度和特异度分别是78.82%和67.63%,均比CA125有所提升。此外本研究中ROMA区别BOT和EOC的AUC为0.822,敏感度为75.0%,特异度为80.4%,结果表明与CA125、HE4单项检测相比,ROMA是BOT和EOC更为有效的生物鉴别诊断工具,很好地平衡了敏感度和特异度。
Fib是调节炎症和癌症发展的关键因素,可介导肿瘤细胞的增殖、迁移和血管生成[39],还与肿瘤细胞免疫逃逸相关,从而促进肿瘤细胞生长[40]。D-d水平可用于早期癌症筛查、监测癌症复发以及评估癌症预后,其对卵巢良恶性肿瘤的鉴别能力也已在其他研究中得到证实[41]。但目前很少应用Fib、D-d对BOT和EOC进行术前鉴别,有研究[42]发现与BOT组相比EOC组的D-d升高概率增加0.3%(OR=1.003,95% CI:1.001-1.006),我们的研究中与BOT组相比EOC组的D-d升高概率增加87.2%(OR=1.872,95% CI:1.361-2.575),这可能与本研究中BOT和EOC样本量较大而他们的样本量不足20例有关。Song 等人研究表明在FAR、预后营养指数(PNI)和SII 3个炎症指标中,FAR在区分卵巢良性肿瘤和卵巢癌方面的敏感性最高,且随着癌症的进展FAR和SII增加PNI减少[42],本研究将FAR应用于BOT和EOC术前鉴别诊断,结果显示EOC组中的FAR水平明显高于BOT组且差异具有统计学意义,但FAR变异性过大,回归方程中对BOT和EOC鉴别效能不确定性增加,今后需进一步扩大样本量。
专家共识推荐进行附件肿块诊断时首选超声检查,至少采用国际卵巢肿瘤分析组织(IOTA)简易标准进行超声特征描述[20]。本研究通过分析BOT组和EOC组的超声特征,进行多因素分析后表明有无乳头、间隔、血流信号是术前鉴别BOT和EOC的独立因素,OR值分别为4.86、9.40、13.84,表明超声特征有助于区分附件肿块的性质。
本研究将BOT和EOC患者的术前血清指标及超声特征联合检测发现,两者联合检测的AUC明显大于血清指标单独检测或仅血清指标联合检测,所有独立影响指标Plt、L、MLR、SII、Fib、ROMA、有无乳头、有无间隔、有无血流信号全部纳入鉴别组合的AUC最大为0.930,敏感度和NPV最高且均大于90%(分别为92.9%、91.9%),ROMA+SII+Fib+MLR+Plt+L+有无血流信号+有无间隔这一组合很好的平衡了鉴别BOT和EOC的敏感度、特异度、PPV和NPV(均为85.7%)。然而Moro等人[43]研究指出BOT与EOC在超声外观上存在重叠,两者都表现为囊肿并伴有乳头状突起,超声检查有时很难评估BOT和EOC,有的病例无法被正确分类,要求超声特征须由经验丰富的妇科超声专家进行评估。
本研究尚存在以下不足:未对BOT和EOC的病理类型进行比较分析,没有进一步量化超声特征中血流评分,且样本来自单中心,需进一步扩大样本量提高研究可信度。
综上所述,一些术前血清指标及超声特征在BOT与EOC之间存在显著差异,术前血清指标ROMA、SII、Fib、MLR、Plt、L联合特殊的超声特征有无乳头、间隔、血流信号可用于BOT和EOC的术前鉴别且具有良好的诊断效能,有助于临床个体化治疗。
1 Kurman RJ,Shih I.The origin and pathogenesis of epithelial ovarian cancer:a proposed unifying theory.Am J Surg Pathol,2010,34:433-443.
2 Torre LA,Trabert B,DeSantis CE,et al.Ovarian cancer statistics,2018.CA Cancer J Clin,2018,68:284-296.
3 Gutiérrez-Casta eda LD,Tovar-Parra D,Quintero G,et al.Isolation and phenotypic characterization of tumor cells of patients with a diagnosis of ovarian cancer.J Cell Physiol,2020,235:3320-3328.
eda LD,Tovar-Parra D,Quintero G,et al.Isolation and phenotypic characterization of tumor cells of patients with a diagnosis of ovarian cancer.J Cell Physiol,2020,235:3320-3328.
4 Peres LC,Cushing-Haugen KL,Köbel M,et al.Invasive epithelial ovarian cancer survival by histotype and disease stage.J Natl Cancer Inst,2019,111:60-68.
5 Jayson GC,Kohn EC,Kitchener HC,et al.Ovarian cancer.Lancet,2014,384:1376-1388.
6 Havrilesky LJ,Sanders GD,Kulasingam S,et al.Development of an ovarian cancer screening decision model that incorporates disease heterogeneity:implications for potential mortality reduction.Cancer,2011,117:545-553.
7 Hauptmann S,Friedrich K,Redline R,et al.Ovarian borderline tumors in the 2014 WHO classification:evolving concepts and diagnostic criteria.Virchows Arch,2017,470:125-142.
8 McCluggage WG,Singh N,Gilks CB.Key changes to the World Health Organization(WHO) classification of female genital tumours introduced in the 5th edition(2020).Histopathology,2022,80:762-778.
9 Wu J,Zhu H,Zhang Q,et al.Nomogram based on the systemic immune-inflammation index for predicting the prognosis of diffuse large B-cell lymphoma.Asia Pac J Clin Oncol,2023,19:e138-e148.
10 Zhang Y,Lin S,Yang X,et al.Prognostic value of pretreatment systemic immune-inflammation index in patients with gastrointestinal cancers.J Cell Physiol,2019,234:5555-5563.
11 Wang Q,Zhu S,Huang X,et al.Prognostic value of systemic immune-inflammation index in patients with urinary system cancers:a meta-analysis.Eur Rev Med Pharmacol Sci,2021,25:1302-1310.
12 Shui Y,Li M,Su J,et al.Prognostic and clinicopathological significance of systemic immune-inflammation index in pancreatic cancer:a meta-analysis of 2,365 patients.Aging(Albany NY),2021,13:20585-20597.
13 Zhang S,Ni Q.Prognostic role of the pretreatment systemic immune-inflammation index in patients with glioma:A meta-analysis.Front Neurol,2023,14:1094364.
14 Borella F,Bertero L,Valabrega G,et al.Searching for prognostic markers for Stage I epithelial ovarian cancer:A role for systemic inflammatory markers.Int J Gynaecol Obstet,2024,164:262-269.
15 Xie H,Yuan G,Liu M,et al.Pretreatment albumin-to-fibrinogen ratio is a promising biomarker for predicting postoperative clinical outcomes in patients with colorectal cancer.Nutr Cancer,2022,74:2896-2909.
16 Holcomb K,Vucetic Z,Miller MC,et al.Human epididymis protein 4 offers superior specificity in the differentiation of benign and malignant adnexal masses in premenopausal women.Am J Obstet Gynecol,2011,205:358.e1-6.
17 许汝福.Logistic回归变量筛选及回归方法选择实例分析.中国循证医学杂志,2016,16:1360-1364.
18 Houck K,Nikrui N,Duska L,et al.Borderline tumors of the ovary:correlation of frozen and permanent histopathologic diagnosis.Obstet Gynecol,2000,95:839-843.
19 Brun J,Cortez A,Rouzier R,et al.Factors influencing the use and accuracy of frozen section diagnosis of epithelial ovarian tumors.Am J Obstet Gynecol,2008,199:244.e1-7.
20 王稳,王兴国,刘淑娟,等.交界性卵巢肿瘤诊治中国专家共识(2022年版).中国实用妇科与产科杂志,2022,38:1185-1194.
21 程英.术中冰冻切片诊断58例卵巢原发性上皮-间质肿瘤的回顾性分析.江西医药,2013,48:72-74.
22 Tempfer CB,Polterauer S,Bentz E,et al.Accuracy of intraoperative frozen section analysis in borderline tumors of the ovary:a retrospective analysis of 96 cases and review of the literature.Gynecol Oncol,2007,107:248-252.
23 Medeiros LR,Rosa DD,Edelweiss MI,et al.Accuracy of frozen-section analysis in the diagnosis of ovarian tumors:a systematic quantitative review.Int J Gynecol Cancer,2005,15:192-202.
24 Rasmussen CB,Kjaer SK,Albieri V,et al.Pelvic inflammatory disease and the risk of ovarian cancer and borderline ovarian tumors:a pooled analysis of 13 case-control studies.Am J Epidemiol,2017,185:8-20.
25 Ramón-Rodríguez J,De-Armas-Conde N,Jaén-Torrejimeno I,et al.Prognostic value of pre-operative systemic immune-inflammation index and platelet to lymphocyte ratio in peritoneal carcinomatosis of ovarian origin.Surg Oncol,2022,42:101750.
26 Gregory AD,Houghton AM.Tumor-associated neutrophils:new targets for cancer therapy.Cancer Res,2011,71:2411-2416.
27 Li Q,Lu Z,Yang L,et al.Neutrophil count and the inflammation-based glasgow prognostic score predict survival in patients with advanced gastric cancer receiving first-line chemotherapy.Asian Pac J Cancer Prev,2014,15:945-950.
28 Li Z,Hong N,Robertson M,et al.Preoperative red cell distribution width and neutrophil-to-lymphocyte ratio predict survival in patients with epithelial ovarian cancer.Sci Rep,2017,7:43001.
29 Xu S,Cao S,Yu Y.High systemic immune-inflammation index is a predictor of poor prognosis in patients with nonsmall cell lung cancer and bone metastasis.J Cancer Res Ther,2021,17:1636-1642.
30 Han R,Tian Z,Jiang Y,et al.Prognostic significance of systemic immune-inflammation index and platelet-albumin-bilirubin grade in patients with pancreatic cancer undergoing radical surgery.Gland Surg,2022,11:576-587.
31 Zhu M,Chen L,Kong X,et al.The systemic immune-inflammation index is an independent predictor of survival in breast cancer patients.Cancer Manag Res,2022,14:775-820.
32 Qi Q,Zhuang L,Shen Y,et al.A novel systemic inflammation response index(SIRI) for predicting the survival of patients with pancreatic cancer after chemotherapy.Cancer,2016,122:2158-2167.
33 Huang H,Wu K,Chen L,et al.Study on the application of systemic inflammation response index and platelet-lymphocyte ratio in ovarian malignant tumors.Int J Gen Med,2021,14:10015-10022.
34 Dikmen ZG,Colak A,Dogan P,et al.Diagnostic performances of CA125,HE4,and ROMA index in ovarian cancer.Eur J Gynaecol Oncol,2015,36:457-462.
35 Gu Z,He Y,Zhang Y,et al.Postprandial increase in serum CA125 as a surrogate biomarker for early diagnosis of ovarian cancer.J Transl Med,2018,16:114.
36 Yanaranop M,Anakrat V,Siricharoenthai S,et al.Is the risk of ovarian malignancy algorithm better than other tests for predicting ovarian malignancy in women with pelvic masses.Gynecol Obstet Invest,2017,82:47-53.
37 Wilailak S,Chan KKL,Chen CA,et al.Distinguishing benign from malignant pelvic mass utilizing an algorithm with HE4,menopausal status,and ultrasound findings.J Gynecol Oncol,2015,26:46-53.
38 Li W,Tang Y,Song Y,et al.Prognostic role of pretreatment plasma d-dimer in patients with solid tumors:a systematic review and meta-analysis.Cell Physiol Biochem,2018,45:1663-1676.
39 Li Y,Yang J,Cheng S,et al.Prognostic significance of FA score based on plasma fibrinogen and serum albumin in patients with epithelial ovarian cancer.Cancer Manag Res,2019,11:7697-7705.
40 Worasethsin P,Narkwichean A.D-dimer as a tumor marker in pre-operative assessment of adnexal masses.J Med Assoc Thai,2013,96:1395-1400.
41 Farzaneh F,Salimnezhad M,Hosseini MS,et al.D-dimer,fibrinogen and tumor marker levels in patients with benign and malignant ovarian tumors.Asian Pac J Cancer Prev,2023,24:4263-4268.
42 Song L,Qi J,Zhao J,et al.Diagnostic value of CA125,HE4,and systemic immune-inflammation index in the preoperative investigation of ovarian masses.Medicine(Baltimore),2023,102:e35240.
43 Moro F,Baima Poma C,Zannoni GF,et al.Imaging in gynecological disease(12):clinical and ultrasound features of invasive and non-invasive malignant serous ovarian tumors.Ultrasound Obstet Gynecol.2017,50:788-799.